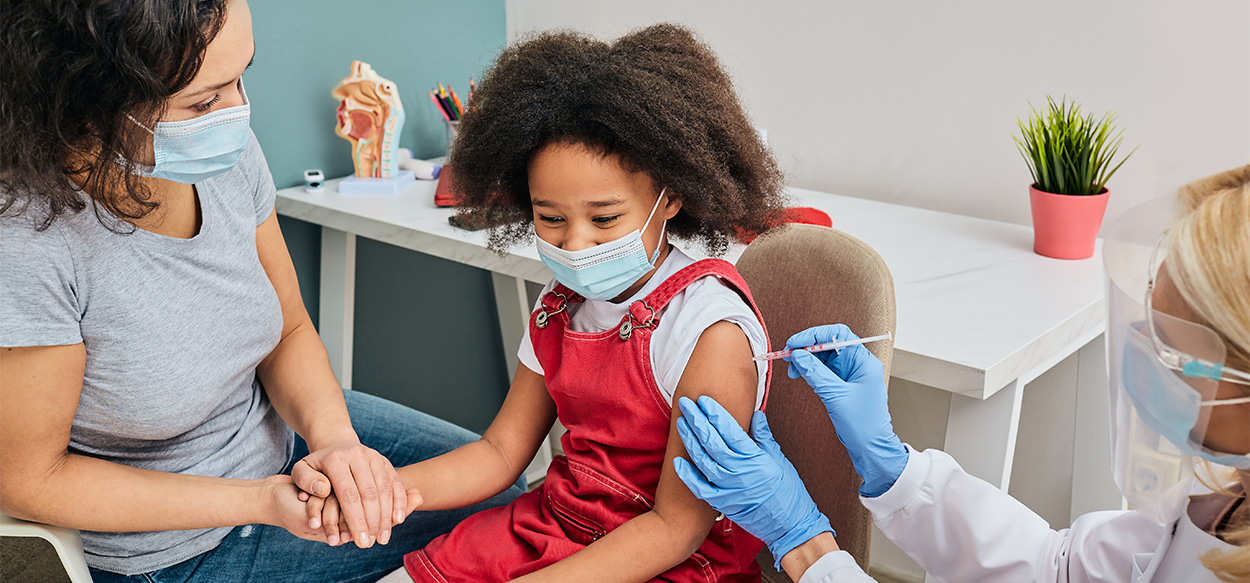
Building Trust One Shot at a Time
Food and Drug Administration (FDA) advisers’ decision to authorize the first COVID-19 vaccine for young children was based, in part, on data from clinical trials conducted by longtime AAHRPP team leader and site visitor Robert Frenck, MD, and his team at Cincinnati Children’s Hospital Medical Division.
Dr. Frenck is director of the Gamble Center for Clinical Research at Cincinnati Children’s and principal investigator of the hospital’s Vaccine Treatment and Evaluation Unit (VTEU). The VTEU is one of five nationwide chosen to help do the initial testing and evaluation of the Pfizer-BioNTech COVID-19 vaccine for children under 12.
The VTEU also has helped assess the AstraZeneca and Pfizer vaccines for adults and, most recently, the Moderna COVID-19 vaccine for pediatric use.
For much of the pandemic, Dr. Frenck’s team worked on-site 60 to 70 hours per week, risking potential coronavirus exposure while “under a microscope every minute. People were nervous, but they put their concerns aside because this is what we trained for. This is our job,” Dr. Frenck says. “In the end, they made history. They’ve been amazing, and I couldn’t be prouder.”
The pressure was greater than at any other time in Dr. Frenck’s more-than-20-year career in vaccine research. “We were watching the world burn, knowing that people were counting on us for vaccine results and hoping for efficacy rates of at least 50% to 60%,” he says.
In November 2020, Dr. Frenck got the call from Pfizer. The results were in and—at 95% efficacy—had exceeded all expectations. “It literally felt like a 10,000-pound weight was lifted off my shoulders,” he says. “All the work had been worth it.”
Pandemic Highs and Lows
That call is at or near the top of Dr. Frenck’s list of pandemic high points. Also up there are nurses at Cincinnati Children’s who were furloughed from their ICU assignments and stepped up to join his vaccine team. Some have since chosen to pursue careers in research.
Individuals, including adolescents and children, who volunteered for his vaccine trials rank high as well. Dr. Frenck speaks of young children who dressed up for the occasion, including a 3-year-old girl who beamed as she announced, “I’m here for my COVID vaccine.” Dr. Frenck’s response: “Good for you!”
Adolescent volunteers impressed him with their enthusiasm and conviction. Almost uniformly, they gave the same answer when he asked why they chose to participate: “If not me, who?” Members of minority populations cited the importance of demonstrating the vaccine’s safety to others in their communities. “Obviously, they wanted to protect themselves,” Dr. Frenck says, “but their primary motivation was to help society.”
On the other hand, vaccine resistance remains a source of frustration, especially “when you work as hard as people have and come up with products as effective as these vaccines. We have a way to stop this pandemic, if only people would get the vaccine.”
If he could deliver just one message, Dr. Frenck would focus on vaccine safety. And that’s exactly what he does in meetings, presentations, and while out in the community, addressing individuals’ concerns one on one while offering them the opportunity to get vaccinated.
“All of these studies were conducted at the highest level, and all regulations were followed. It’s important that we make the community aware of this,” he says. “I am going to keep soldiering on, meeting with people and showing them the data.”
A Matter of Trust
As a pediatrician and professor of pediatrics, Dr. Frenck has seen vaccine hesitancy before but not at the current level. He attributes the intensity both to the highly charged political environment and the need for mass vaccination to fight the pandemic. The situation “has been amplified because you’re asking everyone to get vaccinated at the same time,” he explains.
His solution is rooted in his long-term belief in the importance of building trust—in vaccine development and research in general. That’s one reason Dr. Frenck has been an AAHRPP site visitor since 2007. “We owe the community an assurance that research is being conducted in accordance with the highest standards,” he says, “and AAHRPP accreditation helps provide that.”


